Published March 24, 2015 | Download PDF
The bionic eye is the only retinal prosthesis approved by the U.S. FDA and the first in the world.
(Honolulu, Hawaii, March 24, 2015) The first artificial retinal bionic eye implant surgery in the Asia Pacific region to help restore vision for the blind was performed today in Honolulu, Hawaii by a team at the Eye Surgery Center of Hawaii led by Dr. Gregg Kokame. This ground breaking surgery was performed on March 24, 2015. Dr. Mark Humayun MD PhD, Professor of Ophthalmology at the USC Eye Institute, and the co-inventor of the Argus II implant was also present at the Eye Surgery Center of Hawaii for this historic event.
“Today is a historical day for many reasons,” said Dr. Gregg T. Kokame, founder and president of Retina Consultants of Hawaii and the president of the Eye Surgery Center of Hawaii. “This ‘bionic eye’ implant has the potential to restore vision to patients, who have been in total or near total darkness. This can dramatically change the quality of life of these patients. I have been involved in the care of hereditary blindness since the beginning of my career in Ophthalmology at the UCLA Jules Stein Eye Institute. I have watched patients progressively lose vision, but this new futuristic technology is now available to allow potential vision recovery to patients who are functionally blind.”
The Eye Surgery Center of Hawaii is the leading ophthalmology specialty surgery center focused exclusively on the surgical treatment of the eye in Hawaii and the Asia Pacific. Eighteen eye surgeons perform ophthalmologic surgeries at the Eye Surgery Center of Hawaii. The Eye Surgery Center of Hawaii is a portfolio company of SKAI Ventures, a Hawaii based venture accelerator focused on transforming ingenious ideas into disruptive innovations and high growth companies with global impact.
“We are excited to bring this life-changing technology to Hawaii residents and to the Asia Pacific region,” said Dr. Hank Wuh, CEO of SKAI Ventures. “As the leading center of excellence for vision surgery, we hope to help people with retinal blindness throughout the region.”
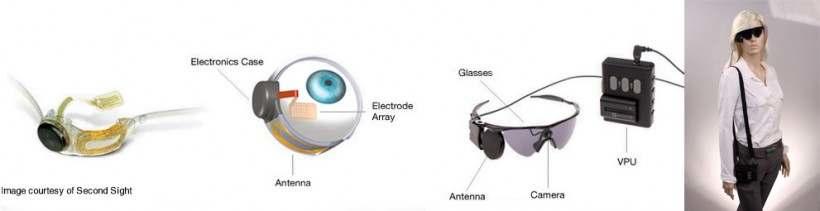
The Argus II (known as the bionic eye) is the only artificial retina that is U.S. FDA approved. The Argus II has shown dramatic clinical results to help patients with retinal blindness be able to utilize artificial vision to see. Today’s implantation of this novel vision restoring device is the first procedure in the Asia Pacific region, and only the second center in the western United States with the first western center being at USC, where the implant was developed.
More artificial retinal bionic implant transplants are scheduled at the Eye Surgery Center of Hawaii. Patients with blindness from retinitis pigmentosa, who are interested in being evaluated for this procedure should contact Dr. Gregg Kokame’s office at 808-487-8928 or email inquiries to research@retinahi.com
Patients from the Asia Pacific region interested in bionic retina transplantation should contact SKAI Ventures Concierge Program at 808-380-8060 or research@retinahi.com.
About Retina Consultants
Retina Consultants of Hawaii (RCH) was founded by Dr. Gregg Kokame. Partners include Dr. Gregg Kokame, Dr. James Lai, and Dr. Raymond Wee. All three ophthalmologists specialize in medical, surgical, and laser treatment of retinal, vitreous, and macular diseases. RCH is globally recognized for patient care, research, and education, and has helped to pioneer many of the leading treatment and technology in use today. Dr. Gregg Kokame is an Iolani graduate, and earned his bachelor’s at Pomona College. He received his MD from UCLA, and completed his ophthalmology residency at the Jules Stein Eye Institute and vitreoretinal surgery fellowship at the Bascom Palmer Eye Institute of the University of Miami, which is ranked the #1 Eye Institute in America for more than a decade. Dr. Kokame is a clinical professor at the University of Hawaii John A. Burns School of Medicine, where he teaches international vitreoretinal fellows, residents, interns and medical students about retinal diseases. All partner physicians at RCH are listed as Best Doctors in Hawaii.
About The Eye Surgery Center of Hawaii (ESCH)
The Eye Surgery Center of Hawaii is the leading state-of-the-art ophthalmology specialty surgery center of excellence focused exclusively on the surgical treatment of eye disease in Hawaii and the Asia Pacific region. Eighteen eye surgeons perform highly specialized ophthalmic surgeries at the Eye Surgery Center of Hawaii. Surgeries performed include treatments for glaucoma, cataract, myopia, retina, cornea, and other eye conditions. All staff and surgical team members are specialists in eye surgery and its subspecialties, allowing the Eye Surgery Center of Hawaii to provide world class expert attention for patient care. The center is able to perform nearly 10,000 surgeries a year.
About SKAI Ventures
SKAI Ventures is a venture accelerator focused on transforming novel, ingenious ideas from physicians, scientists and inventors from around the world, into disruptive innovations and high growth companies with global impact. SKAI Ventures invests in people, technologies, and companies that can positively impact the world and the community we live in. SKAI Ventures actively mentors and nurtures young entrepreneurs and innovators around the globe, especially in Hawaii where the Company was founded. Portfolio companies of SKAI Ventures include the Eye Surgery Center of Hawaii, TruTag Technologies, FitGoFun, Eyegenix, and the Endoscopy Institute of Hawaii.
SKAI’s motto is Invent. Disrupt. Inspire.
About Argus II and Second Sight
The Argus II Retinal Prosthesis System (“Argus II”) is the world’s first approved device intended to restore some functional vision for people suffering from blindness. Argus II is approved for use in the United States (FDA) and European Economic Area (CE Mark) and is available in European countries and the Middle East. Second Sight develops, manufactures and markets implantable visual prosthetics to restore functional vision to blind patients. The company’s current product is an advanced neuro stimulation device called the Argus® II System, which treats retinitis pigmentosa. The Argus II System is the only retinal prosthesis approved in the United States by the Food and Drug Administration and the first approved retinal prosthesis in the world.
Media contact:
Jameson Communications & Strategies
Cell: 808-221-3552
Email: ljameson821@gmail.com or
Jamesonstrategies@gmail.com
Click for High Resolution Photographs:


